News detail
Main Forms of Damage to Mullite Flow Bricks and Factors Affecting Refractoriness
The chemical composition of mullite flow bricks is mainly composed of the mullite phase, with a small amount of low-melting-point iron oxide. The various oxide components in the flow brick have different melting points; if the mass fractions of these components differ, the refractoriness of the flow brick will also differ. The steps for calculating refractoriness using chemical composition are: first, calculate the product of the melting point of each oxide and its corresponding mass fraction; then, sum these products; the result is called the liquidus temperature of the flow brick. Correcting the liquidus temperature allows for estimation of the flow brick’s refractoriness. Simultaneously, the forms and quantities of various complex oxides generated in the reaction layer between the flow brick and molten steel can be inferred.
The types of flow bricks used in ingot casting systems include funnel bricks, pouring tube bricks, center bricks, straight-through bricks, flow tail bricks, and mold bottom bricks. The raw material composition of flowable steel bricks is mainly kaolinite (Al₂O₃·2SiO₂·2H₂O) and 6%~7% impurities (oxides of potassium, sodium, calcium, titanium, and iron). The firing process is mainly the continuous dehydration and decomposition of kaolinite to form mullite (3Al₂O₃·2SiO₂) crystals. Mullite crystals are the only stable form of aluminosilicates under high temperature, with a theoretical conversion rate of 87.64%. During firing, SiO₂ and Al₂O₃ in the flowable steel bricks form eutectic low-melting-point silicates with impurities, surrounding the mullite crystals.
Composition of different batches of steel flow bricks
Test data shows that the main component of steel flow bricks is alumina, followed by silicon dioxide, and also contains small amounts of iron oxide, etc. It is conceivable that the physicochemical properties of steel flow bricks are greatly related to the content of these main components; changes in the main components will have a significant impact on the physicochemical properties of steel flow bricks.
Main Forms of Damage to Refractory Bricks in Steel-Refractory Brick Reaction
When refractory bricks come into contact with molten steel, the interaction between the molten steel and the refractory material includes the following aspects: First, the molten steel washes over the refractory material, causing it to peel off and fall entirely into the molten steel. This is a physical process, which is more likely to occur when there are unstable points such as low-melting-point molten pits in the refractory material. Second, components in the refractory material, such as oxides, carbon, and nitrides, react chemically with elements such as Al, Si, Mn, and Fe in the molten steel. The reaction interface is large, but the thickness of the chemical reaction layer is small. This reaction continues throughout the entire contact process between the refractory material and the molten steel. Since the physical melting process often occurs before the chemical reaction, and the rate of loss due to physical melting is much faster than that of the chemical reaction, the main form of damage to refractory bricks in contact with molten steel can be considered to be physical melting.
The reaction between refractory bricks and molten steel involves physical melting followed by chemical decomposition. The decomposition reaction interface layer is very thin, and the reaction rate is slow. Using the melting point of steel bricks to characterize refractoriness has a certain scientific basis, and the calculation method is relatively simple.
Calculation Process of Chemical Composition
3.1 Chemical Composition of a Batch of Steel Flow Bricks
After a batch of steel flow bricks arrived at the factory, samples were taken to test their chemical composition.
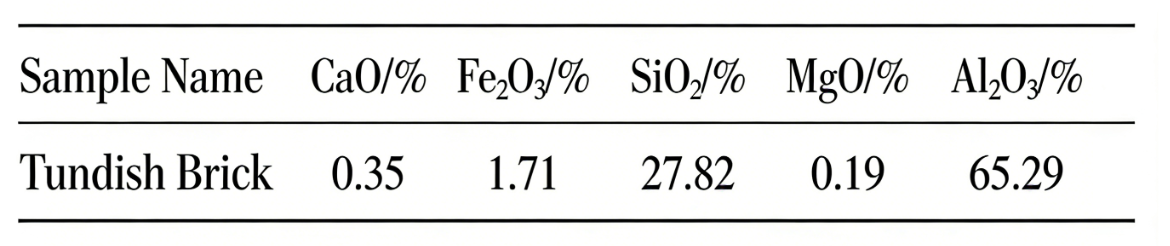
As shown in Table 1, the chemical composition is relatively normal, with significant differences in the mass fractions of the main and minor components.
3.2 Calculation of the Liquidus of a Batch of Flow Steel Bricks
The formula for calculating the number of oxide molecules MxOy is:
aMxOy=XMxOy/AMxOy (1)
Where: aMxOy is the number of oxide molecules MxOy; XMxOy is the mass fraction of oxide MxOy, %; AMxOy is the molecular weight of oxide MxOy.
During the contact process between the flow steel brick reaction layer and the high-temperature molten steel, the oxides in the reaction layer always tend to undergo a combination reaction with the lower melting point phase.
When only Al₂O₃, SiO₂, CaO, MgO, and Fe₂O₃ are calculated, the liquidus of the flow steel brick with an Al₂O₃ content of 65.29% is 1854℃. Besides the mullite-forming oxides such as Al₂O₃ and SiO₂, the high-alumina bauxite used in the raw material of flowable bricks also contains a certain amount of impurities. Among these impurities, Fe₂O₃, K₂O, and Na₂O have low melting points. During the contact between molten steel and the refractory surface, the high-temperature molten steel easily dissolves these impurities on the refractory surface, forming liquid channels that penetrate into the interior of the refractory. Therefore, these low-melting-point oxides reduce the refractoriness of the flowable bricks. The actual liquidus temperature of a flowable brick with an Al₂O₃ content of 65.29% should be below 1854℃.
Composition of Mineral Phases Formed in Flowing Steel Bricks During High-Temperature Reactions
From a thermodynamic perspective, during the contact between molten steel and flowing steel bricks, various oxides in the flowing steel bricks tend to combine with lower-melting-point complex oxides. During the reaction between the flowing steel bricks and high-temperature molten steel, complex compounds are continuously generated in the reaction layer of the flowing steel bricks. When the reaction temperature of the molten steel exceeds the melting point of the complex oxides, some complex oxide liquid phases are formed in the reaction layer of the flowing steel bricks. These liquid phases of complex oxides are formed at the reaction interface layer and are drawn into the flowing molten steel. Following the principle of preferential formation of lower-melting-point complex oxides, and based on the molecular weight of each oxide, the molecular formulas and quantities of various complex compounds in the reaction layer of the flowing steel bricks can be deduced.
There are 46 SiO₂ molecules, 64 Al₂O₃ molecules, and only 1 CaO and 1 Fe₂O₃ molecule each. Therefore, SiO₂ and Al₂O₃ will preferentially combine. The most preferentially formed complex compound in Table 6 is mullite, followed by calcium aluminate, both of which have very low melting points. Fe₂O₃ has the lowest melting point, but a single Fe₂O₃ molecule will still exist as a separate oxide. In 3Al₂O₃·2SiO₂, the ratio of Al₂O₃ to SiO₂ is 3:2, so approximately 64:43 of Al₂O₃ and SiO₂ form mullite, leaving 3 SiO₂ molecules. There is only one CaO, one Fe₂O₃, and one CaO, one Al₂O₃ and one SiO₂ form one CaO·FeO·SiO₂, leaving two SiO₂ atoms.
Conclusion
(1) The main damage occurring during the contact between the steel flow brick and molten steel is a physical melting process. The decomposition reaction interface layer of oxides is relatively thin, resulting in a slow reaction rate. The melting point and corresponding mass fraction of various oxides in the steel flow brick jointly determine the liquidus temperature of the reaction between the steel flow brick and molten steel.
(2) When the steel flow brick reacts with the molten steel at the interface, the reaction occurs by the formation of a single oxide complex from multiple individual oxides. The lower the melting point of the complex oxide, the greater the reaction kinetics for forming this complex oxide; the higher the mass fraction of the individual oxide, the easier it is for this oxide to undergo a combination reaction.
(3) The method of deriving refractoriness using chemical composition calculations involves calculating an accurate liquidus temperature value using the mass fraction and melting point of each component.


Send inquiry
Please Leave your message you want to know! We will respond to your inquiry within 24 hours!